Dear Greg and David:
Thank you for your comments on our 2009 paper (Fedorenko & Kanwisher, 2009, Language and Linguistics Compass). As you stated in your post on August 24, 2010, and as became even clearer from the email exchange that took place the following day, you agree with Nancy Kanwisher and me on the importance of taking into consideration anatomical and functional variability in fMRI research in any domain, including language. This was the main point of our article, so on the important points we agree. We will here focus on the points where there is some disagreement between us.
1. How much do we, as a field, know about functional specialization of language-sensitive brain regions?
Although different researchers in the fields of psycho- and neuro-linguistics may vary in how satisfied they are with the progress that has been made in understanding the brain basis of language, we don't think anyone would argue that we have arrived at a complete understanding of what computations each language-sensitive brain region performs and/or what representations it stores/manipulates. So, given that we can probably agree on this, the question becomes: how much do we actually know, as a field, about the brain basis of language in general, and about the questions of functional specialization of language-sensitive brain regions, in particular.
As you point out, we have argued that two fundamental questions concerning the functional architecture of the language system remain unanswered:
• Do language-sensitive brain regions each support one particular aspect of language, or do they support multiple linguistic processes?
• Do language-sensitive regions specialize for (some aspect of) language processing, or do they also support some non-linguistic processes (e.g., general working memory, arithmetic processing, music, action representation, etc.)?
You implied that our pessimistic assessment is due to ignorance. In fact, we are aware of the relevant literature, but we think that the findings that you refer to, among others, either do not pertain to or conclusively answer the questions of functional specificity.
Let us first clarify what we mean by “functional specificity”, because perhaps some of the confusion stems from different interpretations of the issues at hand. Here is our working definition: A brain region R is specialized for cognitive function x if this region (i) is engaged in tasks that rely on cognitive function x, and (ii) is not engaged in tasks that do not rely on cognitive function x.
It is important to clarify the difference between a brain region supporting a particular cognitive function and a brain region selectively supporting that cognitive function. A demonstration of a relationship between brain region R and cognitive function x tells us little about R’s specificity for x. For example, various working memory tasks consistently activate dorsolateral prefrontal cortical regions, across studies and labs. However, this does not mean that these regions are selectively engaged in working memory; instead, these regions appear to support a wide range of demanding cognitive tasks (e.g., Duncan, 2001, Nat Rev Neurosci Duncan, 2010, TiCS). We elaborate on this issue – with respect to the claim that a portion of Broca’s area is selectively engaged in one aspect of syntax (e.g., Grodzinsky & Santi, 2008, TiCS) – in our response to Grodzinsky (Fedorenko & Kanwisher, submitted; see also Willems & Hagoort, 2009, TiCS, for similar arguments).
Now, let’s turn to your claims.
Concerning within-language specificity, you said:
“… there is strong evidence from functional imaging regarding the involvement of distinct cortical regions/circuits in phonemic (STS), lexical-semantic (MTG), prosodic (anterior dorsal STG), and higher-level combinatorial processes (anterior temporal/inferior frontal regions). Additional circuits have been delineated that support auditory-motor integration and auditory/phonological short-term memory. Here are some relevant reviews of this literature: Binder et al., 2000; Hickok and Poeppel, 2007; Indefrey and Levelt, 2004.”
Concerning domain specificity of language-sensitive regions, you said:
“Regarding question two, several studies have clearly identified voice-specific responses in the STG (Belin et al. 2000), higher-level speech specific responses in the STS (Scott et al. 2000; Okada et al. 2010), and even what we might as well call the anterior temporal lobe sentence area, given how selective it is to the perception of sentence-level stimulation (Humphries et al., 2006; Humphries et al., 2005; Humphries et al., 2001; Rogalsky and Hickok, 2009; Vandenberghe et al., 2002). But more importantly, some of us have moved beyond the specificity issue with the aim of trying to identify the circuits and computations involved in a given process whether or not it is special to speech.”
Then later you pointed out that we have not cited several important findings from the recent literature. Of relevance to the questions of functional specificity are the following (NB: (i) and (iii) are not directly relevant to this question, so we will not discuss those here):
“(ii) convergence on the involvement of the STS in phonemic level processes in speech perception (Leibenthal et al., 2005; Scott & Johnsrude, 2003; Hickok & Poeppel, 2007)”;
“(iv) recent progress in mapping the circuit that supports sensory-motor integration in speech processing (Golfinopoulus et al. 2009; Hickok et al., 2009)”;
“(v) progress in understanding the basis of hemispheric asymmetries for acoustic and phonemic processing in auditory cortex (Boemio, et al. 2005); Zatorre, et al. 2002)
“(vi) convergence on the idea that anterior temporal regions support some aspect of sentence-level processing (the linguistic equivalent of the FFA)”;
“(vii) convergence on the relation between sensory-motor circuits and phonological short-term memory (Buchsbaum et al. 2008; Postle, 2006)”.
Although we are familiar with the findings and papers you mention, we don’t think that – with respect to any of the brain regions above – we can conclusively say: this region exclusively performs cognitive function x, or cognitive functions x1-xn (if a region is truly multifunctional). This is not to say that some of the studies you mention, as well as dozens of other studies, have not narrowed down the space of possible hypotheses for the function(s) of various regions.
Let’s consider one of your examples above: the claim that “anterior temporal regions support some aspect of sentence-level processing”; this area, according to you, is “selective […] to the perception of sentence-level stimulation”, and you even go as far as calling it “the linguistic equivalent of the FFA”. The papers you cite in support of this claim indeed quite strongly suggest that this region is engaged in processing sentence-level material. However, in order to argue that this region selectively supports sentence-level understanding, more work is needed. In particular, language has been argued to share various properties with a number of non-language tasks (e.g., arithmetic, musical processing, action representation, general executive functions, aspects of social cognition, etc.). Furthermore, tasks in some of these domains have been shown to activate regions in/around anterior temporal cortex (just to provide a few examples: amodal conceptual processing – e.g., Patterson et al., 2007, Nat Rev Neurosci; memory for music – e.g., Peretz, 1996, J of Cog Neurosci; aspects of social cognition – e.g., Zahn et al., 2007, PNAS; Ross & Olson, 2010, Neuroimage; face processing – e.g., Sugiura et al., 2001, Neuroimage). Perhaps these different non-language tasks activate different portions of the ATL than sentence-level understanding, and perhaps you can dismiss some of these claims / findings on the basis of some design, methodological or analysis flaws. Nevertheless, in order to convince the world that anterior temporal regions are specialized for some aspect of sentence-level processing (as you seem to argue), you would have to show that those regions do not respond to any of the tasks that have been shown to activate regions in/around ATL (as e.g., Kanwisher and colleagues have done for the FFA and other ventral visual stream regions).
Another finding you mention is “the involvement of the STS in phonemic level processes in speech perception”. As with the role of the ATL structures in sentence-level understanding, we agree with you that there is strong evidence for the engagement of some portions of STS in phonemic level processes. Perhaps this is even the strongest case of functional specialization in language-related brain regions since across several studies (including the ones you cite) a stronger response to speech sounds has been demonstrated relative to several non-speech auditory control conditions (cf. Leech et al., 2009, J Neurosci, for a claim that speech-sound-sensitive areas in STS are not domain specific but rather subserve the processing of acoustically complex sounds more generally and only show higher responses to speech sounds due to more extensive experience with those sounds). However, as with the ATL structures, we think more work is needed. Within language, some studies have argued for the sensitivity of STS regions (including the posterior portion of it) to high-level linguistic manipulations (see e.g., Caplan, 2007, Language & Linguistics Compass, for a review of some such studies; see also Friederici et al., 2010, Hum Brain Mapping, among others). Furthermore, a number of non-linguistic tasks have been shown to activate regions in/around STS, such as action representation (e.g., Vander Wyk et al., 2009, Psych Sci) or aspects of social cognition (e.g., Allison et al., 2000, TiCS), among others (see Hein & Knight, 2008, J Cog Neurosci, for a recent review). Again, as with the ATL structures, these different tasks may activate regions that are non-overlapping with the speech-sound-sensitive regions. However, this needs to be demonstrated empirically.
In summary, in order to make claims about functional specificity of some region R for a cognitive function x, it is necessary to examine R’s response to a wide range of linguistic and non-linguistic tasks that have either been (a) argued to share some properties with x on theoretical grounds, and/or (b) shown to activate cortex in/around R. (Group-based methods can be used for addressing these questions, but individual-subject analyses are particularly well suited as they will be most likely to uncover dissociations if such are present (Nieto-Castanon, Fedorenko & Kanwisher, in prep.))
Nevertheless, despite the fact that questions of functional specificity have not yet been “nailed” for any of the language-sensitive regions, you are right that previous work has contributed significantly to narrowing down the space of possible hypotheses for the functions of many of these regions (and perhaps this is what you wanted us to acknowledge more in our 2009 paper). Our 2009 paper was not intended to be a complete review of the relevant literature. Instead, our approach in that paper (as well as in the 2010 J Neurophys paper) has been to fairly broadly state the research questions that, in our opinion, remain open as of yet (i.e., the questions of functional specificity of language-sensitive regions). This approach has its limitations: some researchers may feel neglected and/or not properly acknowledged for their contributions to the field. We apologize for any such omissions.
2. Understanding the degree of functional specialization of a region is critical to discerning the computations it supports and/or the representations it stores/manipulates.
You said:
“But more importantly, some of us have moved beyond the specificity issue with the aim of trying to identify the circuits and computations involved in a given process whether or not it is special to speech.”
We don't understand this assertion. We also would like to understand the computations that different language-sensitive brain regions perform. However, understanding whether / how specialized a particular region is for language (or a particular aspect of linguistic processing), is essential for understanding the region's computations, so the juxtaposition is confusing.
There are at least three possible research strategies for discerning the nature of the computations/representations in language-sensitive brain regions. One could start by characterizing the language system (i.e., all the language-sensitive brain regions) in terms of their within-language specificity: figuring out whether different regions specialize for particular aspects of language vs. support multiple aspects of language, and understanding how these regions work together to enable language production /comprehension. Then one could use this information to further examine whether these regions support any non-linguistic processes. Alternatively, one could start by characterizing language-sensitive brain regions in terms of their domain specificity: figuring out which, if any, non-linguistic cognitive processes different language-sensitive regions support. Then one could use this information to further examine the precise role of these regions in linguistic processing. Finally, one could simultaneously tackle both questions – (i) within-language specificity, and (ii) domain specificity – and treat the findings with respect to these questions as mutually informative. We are adopting the third approach and hope to use knowledge accumulated in both of these lines of research to draw inferences about the function(s) of each language-sensitive region.
(Of course, fMRI is also not the only method that is needed to answer these questions definitively: high-temporal-resolution methods will be critical for potentially dissociating computations that may be performed by the same cortical tissue but occur at different time-scales.)
Somewhat relatedly, in another one of your posts (http://www.talkingbrains.org/2008/09/brodmann-areas-and-localization-in.html), you said the following:
“Ted Jones provides an instructive reminder in this new age of localization-based neuroscience: “No cortical area is an isolated entity in which a single function is represented. Nor, contrary to many current views, does it merely form one step in a hierarchy of areas proceeding onwards and upwards to some defined or imagined higher function. While there are definite streams of cortico–cortical connections that proceed in identifiable ways from area to area in the cortex, no area is without feedback connections and no area is without re-entrant connections from the thalamus.””
Although it is certainly true that any given brain region is connected to numerous other brain regions, we do not agree with the assertion that “no cortical area is an […] entity in which a single function is represented”. Several regions in the ventral visual stream have been shown to be exquisitely specialized for processing visual stimuli of a particular class (see e.g., Kanwisher, 2010, PNAS, for a recent overview). Furthermore, Saxe and colleagues have shown that a region in the right temporo-parietal junction selectively responds to stimuli that require us to think about what another person is thinking (e.g., Saxe & Powell, 2006, Psych Sci, and many other papers; see the publications section on the SaxeLab’s website: http://saxelab.mit.edu/publications.php). It is possible that none of the language-sensitive brain regions will show this degree of functional specialization. However, this question deserves to be investigated in the most rigorous way possible given its centrality to understanding human cognition in general.
Given numerous reports of what look like highly specialized linguistic deficits following focal brain damage, we tend to think that at least some degree of functional specialization is bound to be present in the language system. However, we are not on a quest to demonstrate functional specialization of some brain region for language (or for a particular aspect of language). Instead, we want to understand the functional response profiles of different language-sensitive brain regions, and the methods we advocate are perfectly suited for this task. Only by understanding how a region responds to a wide range of cognitive tasks can we begin to make inferences about the computations it performs and the representations that it stores/manipulates.
3. A couple of comments on “methodological schooling”.
You said:
“This is frankly a pathetic summary of the state of the field and a pretentious starting point for the methodological schooling that F&K provide in the following sections of their paper.”
“Methodological schooling” is not our intention. A few points are worth making here.
a) Many brain imaging methods have a place in investigations of the brain basis of language.
We tried to make this clear in our 2010 paper and in our response to Grodzinsky (Fedorenko & Kanwisher, submitted) and will keep trying to get this point across in our future talks and papers: we strongly believe that individual-subject analyses (both the “basic” subject-specific fROI analyses as well as some more sophisticated individual-subject analyses we’ve been developing) can go a long way in providing clearer answers to some outstanding questions. However, we are not arguing for abandoning the traditional, group-based, methods (or for ignoring the knowledge we have gained from those methods). Rather we think that a multi-pronged approach to the study of language – using all the tools available to us – is what’s needed for faster progress in the field.
b) How novel are functional localizers in language research?
We do realize and explicitly acknowledge (e.g., Fedorenko et al., 2010, J Neurophys, p. 1178) that some researchers have used individual subject analyses previously (we apologize for not including your studies in this list; we will do that in the future). However, as we note in the paper, to the best of our knowledge, prior to our 2010 paper (and efforts in the clinical literature to localize language-sensitive cortex using fMRI, which have not been adopted in the non-clinical literature), no contrast aimed at localizing language-sensitive brain regions has been validated, i.e., shown to pick out the same regions reliably within and across subjects. Without independent validation of the functional localizer contrasts, it may be difficult to interpret the responses of the ROIs to the critical condition of interest. What are needed are not “ad hoc” localizers that are different across different studies / labs but standardized ones that have been shown to effectively identify the target regions. For example, the localizer that you used in Rogalsky & Hickok (2009, Cereb Cortex) – a contrast between sentences and word lists – identifies only a region in the ATL and does not find any posterior temporal or frontal regions, so does not manage to capture many regions known to be engaged in sentence processing. A key goal of our 2010 J Neurophys paper was to test and establish the robustness of a specific localizer that successfully identifies most of these regions and hence will be of widespread use for many future studies. (Note that although we use the contrast between sentences and nonwords as our main contrast, the contrast between sentences and word lists in our localizer identifies very similar regions to those identified with the sentences > nonword lists contrast, including left frontal and temporal / temporo-parietal regions.)
Another contribution of our work is in developing a method for objectively and efficiently delineating the borders of functional ROIs in individual subjects, the group-constrained subject-specific (GSS) method (for more info see: http://web.mit.edu/evelina9/www/funcloc.html; see Julian et al., in prep., for validation of this method on well characterized high-level visual regions).
c) We invite others to try our methods.
As we mentioned in an email to you, by developing a way to quickly and reliably identify language-sensitive regions, we are not trying to say, “You have all done this wrong; see how we can do it better", but rather, we want to relay something along the lines of, “There may be some limitations in the methods that are currently in use in the vast majority of fMRI studies of language; here is a new approach: let's see how we, as a field, can put our heads together and harness the power of individual subject analyses – by developing a series of standard language localizers and using them consistently across studies and labs – to bring more clarity to where it's needed”.
By making all our tools publicly available (http://web.mit.edu/evelina9/www/funcloc.html), we hope that other researchers will join us in adopting the subject-specific approach and that, as a field, we can develop a set of standard localizers for various aspects of language that can be used consistently across studies and labs. We can then – in a joint effort – systematically characterize a set of key brain regions that enable proper linguistic functioning by examining their response to a wide range of linguistic and non-linguistic stimuli and tasks, thus deriving detailed functional profiles for each region.
We would like to conclude by commenting on the following assertion you make at the end of your post:
“individual subject analyses would be helpful, but it is not a magic bullet (e.g., task selection is more important in my view)”
First, we agree that task selection is highly important in characterizing functional profiles of language-sensitive (or other) regions. However, we don’t have to choose between good task design and the use of ROIs: we can do both.
Second, we are advocating individual subject analyses, and we strongly believe that such analyses will bring a clearer understanding of the brain basis of language. We have not shown this yet, so there is no point to argue against this. So far, we developed a method and several new analysis tools that now enable us to pursue questions of functional specificity with rigor that has not so far been possible, because (i) group-based methods are guaranteed to underestimate specificity, and (ii) without a standard localizer used to define ROIs across studies / labs, it is difficult to compare activations across studies / labs, because one can never – with certainty – determine whether an activation peak in one study reflects the activity of the same region as the activation peak in another study, or of a different / nearby region. Of course, it remains to be seen what this method can bring to the field in terms of our understanding of the language architecture. We think it's too early to make pronouncements like the one you make above. Why not give this method a chance? We invite you and others in the field to join our efforts.
Best,
Ev Fedorenko and Nancy Kanwisher
P.S. Our response to Grodzinsky’s critique of our 2009 paper will soon be available from Ev’s website (http://web.mit.edu/evelina9/www/).
P.P.S. One last comment – not related to the issues above – remains to be made. You made a parallel between the current debate and another debate that I (Ev) was recently involved in: the debate about whether quantitative methods are needed in linguistics research (Gibson & Fedorenko, 2010, TiCS; Gibson & Fedorenko, in press, LCP). These two debates are quite different, in spite of some superficial similarities. Unlike brain imaging research on language, where a choice of an analysis method may be driven by the nature of the research question and sometimes by researchers’ preferences, the use of quantitative methods in linguistics it is not a matter of a research question or preference/opinion. Quantitative methods should be used in linguistics, as they are used in every other branch of cognitive science, and science more generally.
News and views on the neural organization of language moderated by Greg Hickok and David Poeppel
Tuesday, August 31, 2010
Tuesday, August 24, 2010
Do you like movies? MEG? Multi-sensory perception?
Doing our best to keep this rather technical and slightly nerdy paper light (e.g. the stimuli were clips from the film Dumb and Dumber), this new paper in PLoS Biology by Huan Luo, Zuxiang Liu, David Poeppel argues for active cross-modal phase modulation as a key mechanism underlying the construction of internal representations of naturalistic audiovisual stimuli. The paper builds on a technique that Huan Luo developed for an earlier paper (Luo & Poeppel, 2007, Neuron) that argues for the relevance of theta phase in auditory cortex. If you are looking for something to read during the last days of your summer vacation ...
Auditory Cortex Tracks Both Auditory and Visual Stimulus Dynamics Using Low-Frequency Neuronal Phase Modulation
'Low-key' author summary for PLoS:
When faced with ecologically relevant stimuli in natural scenes, our brains need to coordinate information from multiple sensory systems in order to create accurate internal representations of the outside world. Unfortunately, we currently have little information about the neuronal mechanisms for this cross-modal processing during online sensory perception under natural conditions. Neurophysiological and human imaging studies are increasingly exploring the response properties elicited by natural scenes. In this study, we recorded magnetoencephalography (MEG) data from participants viewing audiovisual movie clips. We developed a phase coherence analysis technique that captures—in single trials of watching a movie—how the phase of cortical responses is tightly coupled to key aspects of stimulus dynamics. Remarkably, auditory cortex not only tracks auditory stimulus dynamics but also reflects dynamic aspects of the visual signal. Similarly, visual cortex mainly follows the visual properties of a stimulus, but also shows sensitivity to the auditory aspects of a scene. The critical finding is that cross-modal phase modulation appears to lie at the basis of this integrative processing. Continuous cross-modal phase modulation may permit the internal construction of behaviorally relevant stimuli. Our work therefore contributes to the understanding of how multi-sensory information is analyzed and represented in the human brain.
Auditory Cortex Tracks Both Auditory and Visual Stimulus Dynamics Using Low-Frequency Neuronal Phase Modulation
'Low-key' author summary for PLoS:
When faced with ecologically relevant stimuli in natural scenes, our brains need to coordinate information from multiple sensory systems in order to create accurate internal representations of the outside world. Unfortunately, we currently have little information about the neuronal mechanisms for this cross-modal processing during online sensory perception under natural conditions. Neurophysiological and human imaging studies are increasingly exploring the response properties elicited by natural scenes. In this study, we recorded magnetoencephalography (MEG) data from participants viewing audiovisual movie clips. We developed a phase coherence analysis technique that captures—in single trials of watching a movie—how the phase of cortical responses is tightly coupled to key aspects of stimulus dynamics. Remarkably, auditory cortex not only tracks auditory stimulus dynamics but also reflects dynamic aspects of the visual signal. Similarly, visual cortex mainly follows the visual properties of a stimulus, but also shows sensitivity to the auditory aspects of a scene. The critical finding is that cross-modal phase modulation appears to lie at the basis of this integrative processing. Continuous cross-modal phase modulation may permit the internal construction of behaviorally relevant stimuli. Our work therefore contributes to the understanding of how multi-sensory information is analyzed and represented in the human brain.
Neuroimaging of language: Why hasn't a clearer picture emerged?
This is the question raised in a paper by Evelina Fedorenko and Nancy Kanwisher published last year in Language and Linguistics Compass. The main point that they want to make is that language neuroimagers need to stop doing group studies and start doing functional localization in individual subjects, like the vision folks do. I don't disagree at all; e.g., see this post. In fact, we have used individual subject analyses in several of our papers (e.g., Okada & Hickok, 2006; Okada et al., in press).
What I found a bit on the irritating side though was the extremely dim and distressingly myopic view of progress in the field of the neural basis of language. They start by stating that two questions have
And they suggest that "Neuroimaging has not yet provided clear answers to either question".
Regarding question one, there is strong evidence from functional imaging regarding the involvement of distinct cortical regions/circuits in phonemic (STS), lexical-semantic (MTG), prosodic (anterior dorsal STG), and higher-level combinatorial processes (anterior temporal/inferior frontal regions). Additional circuits have been delineated that support auditory-motor integration and auditory/phonological short-term memory. Here are some relevant reviews of this literature: Binder et al., 2000; Hickok and Poeppel, 2007; Indefrey and Levelt, 2004.
Regarding question two, several studies have clearly identified voice-specific responses in the STG (Belin et al. 2000), higher-level speech specific responses in the STS (Scott et al. 2000; Okada et al. 2010), and even what we might as well call the anterior temporal lobe sentence area, given how selective it is to the perception of sentence-level stimulation (Humphries et al., 2006; Humphries et al., 2005; Humphries et al., 2001; Rogalsky and Hickok, 2009; Vandenberghe et al., 2002). But more importantly, some of us have moved beyond the specificity issue with the aim of trying to identify the circuits and computations involved in a given process whether or not it is special to speech.
So F&K are a bit misinformed in regarding the contribution of neuroimaging to the questions they raise.
Even worse, though, is their summary of "where things stand" concerning our understanding of the "neural basis of language" -- a rather sweeping domain, especially if they have in mind only the two questions they raise at the outset. Nonetheless, concerning the Neural Basis of Language they emphasize:
1. that the 19th century idea that Broca's area = speech production and Wernicke's area = speech comprehension doesn't hold up to modern data
2. that left frontal regions activate to a variety of language tasks, and indeed even non-linguistic tasks
3. that regions outside of the traditional peri-Sylvian cortex activate during language processing
4. that meta analyses show lots of overlap between language tasks
This is frankly a pathetic summary of the state of the field and a pretentious starting point for the methodological schooling that F&K provide in the following sections of their paper.
Completely ignored in this summary is (i) a body of work showing that much of the confusion (and overlapping activations) evaporates if one is careful about task selection (Hickok & Poeppel, 2007), (ii) convergence on the involvement of the STS in phonemic level processes in speech perception (Leibenthal et al., 2005; Scott & Johnsrude, 2003; Hickok & Poeppel, 2007), (iii) convergence on the idea of a dual stream architecture in language system (Hickok & Poeppel, 2007; Rauchecker & Scott, 2009), (iv) recent progress in mapping the circuit that supports sensory-motor integration in speech processing (Golfinopoulus et al. 2009; Hickok et al., 2009), (v) progress in understanding the basis of hemispheric asymmetries for acoustic and phonemic processing in auditory cortex (Boemio, et al. 2005); Zatorre, et al. 2002) , (vi) convergence on the idea that anterior temporal regions support some aspect of sentence-level processing (the linguistic equivalent of the FFA), (vii) convergence on the relation between sensory-motor circuits and phonological short-term memory (Buchsbaum et al. 2008; Postle, 2006)... I could go on.
Yes, there is still plenty of murkiness, much of it surrounding the function of Broca's area, and yes, individual subject analyses would be helpful, but it is not a magic bullet (e.g., task selection is more important in my view) -- e.g., I'm willing to bet that the vision folks still have some work to do -- and the existence of murkiness doesn't justify the characterization of an entire field as failing to make progress due to methodological ineptness. This kind of argumentation was prominent in another of Fedorencko's papers that was featured prominently on this blog. It's a bit disturbing to see it showing up again.
F&K's paper has generated a more formal (i.e., published) response by Grodzinsky (2010) who is critical of their take on the field as well but for different reasons. Definitely worth a look.
The field of the neural basis of language has made significant progress in the last several years, despite what F&K assert.
References
Fedorenko, E., & Kanwisher, N. (2009). Neuroimaging of Language: Why Hasn't a Clearer Picture Emerged? Language and Linguistics Compass, 3 (4), 839-865 DOI: 10.1111/j.1749-818X.2009.00143.x
Binder, J.R., Frost, J.A., Hammeke, T.A., Bellgowan, P.S., Springer, J.A., Kaufman, J.N., and Possing, E.T. (2000). Human temporal lobe activation by speech and nonspeech sounds. Cerebral Cortex 10, 512-528.
Belin, P., Zatorre, R.J., Lafaille, P., Ahad, P., and Pike, B. (2000). Voice-selective areas in human auditory cortex. Nature 403, 309-312.
Buchsbaum, B.R., and D'Esposito, M. (2008). The search for the phonological store: from loop to convolution. J Cogn Neurosci 20, 762-778.
Golfinopoulos, E., Tourville, J.A., and Guenther, F.H. (2009). The integration of large-scale neural network modeling and functional brain imaging in speech motor control. Neuroimage.
Grodzinsky, Yosef. 2010. A clearer view of the linguistic brain: reply to Fedorenko and Kanwisher. Language and Linguistic Compass, 4, pp. 605-622.
Hickok, G., and Poeppel, D. (2007). The cortical organization of speech processing. Nat Rev Neurosci 8, 393-402.
Humphries, C., Binder, J.R., Medler, D.A., and Liebenthal, E. (2006). Syntactic and semantic modulation of neural activity during auditory sentence comprehension. J Cogn Neurosci 18, 665-679.
Humphries, C., Love, T., Swinney, D., and Hickok, G. (2005). Response of anterior temporal cortex to syntactic and prosodic manipulations during sentence processing. Human Brain Mapping 26, 128-138.
Humphries, C., Willard, K., Buchsbaum, B., and Hickok, G. (2001). Role of anterior temporal cortex in auditory sentence comprehension: An fMRI study. Neuroreport 12, 1749-1752.
Indefrey, P., and Levelt, W.J. (2004). The spatial and temporal signatures of word production components. Cognition 92, 101-144.
Liebenthal, E., Binder, J.R., Spitzer, S.M., Possing, E.T., and Medler, D.A. (2005). Neural substrates of phonemic perception. Cereb Cortex 15, 1621-1631.
Okada, K., and Hickok, G. (2006). Identification of lexical-phonological networks in the superior temporal sulcus using fMRI. Neuroreport 17, 1293-1296.
Okada, K., Rong, F., Venezia, J., Matchin, W., Hsieh, I.H., Saberi, K., Serences, J.T., and Hickok, G. (in press). Hierarchical Organization of Human Auditory Cortex: Evidence from Acoustic Invariance in the Response to Intelligible Speech. Cereb Cortex.
Postle, B.R. (2006). Working memory as an emergent property of the mind and brain. Neuroscience 139, 23-38.
Rauschecker, J.P., and Scott, S.K. (2009). Maps and streams in the auditory cortex: nonhuman primates illuminate human speech processing. Nat Neurosci 12, 718-724.
Rogalsky, C., and Hickok, G. (2009). Selective Attention to Semantic and Syntactic Features Modulates Sentence Processing Networks in Anterior Temporal Cortex. Cereb Cortex 19, 786-796.
Scott, S.K., and Johnsrude, I.S. (2003). The neuroanatomical and functional organization of speech perception. Trends Neurosci 26, 100-107.
Scott, S.K., Blank, C.C., Rosen, S., and Wise, R.J.S. (2000). Identification of a pathway for intelligible speech in the left temporal lobe. Brain 123, 2400-2406.
Vandenberghe, R., Nobre, A.C., and Price, C.J. (2002). The response of left temporal cortex to sentences. J Cogn Neurosci 14, 550-560.
What I found a bit on the irritating side though was the extremely dim and distressingly myopic view of progress in the field of the neural basis of language. They start by stating that two questions have
driven dozens of studies on the neural basis of language published in the last several decades: (i) Are distinct cortical regions engaged in different aspects of language? (ii) Are regions engaged in language processing specific to the domain of language?
And they suggest that "Neuroimaging has not yet provided clear answers to either question".
Regarding question one, there is strong evidence from functional imaging regarding the involvement of distinct cortical regions/circuits in phonemic (STS), lexical-semantic (MTG), prosodic (anterior dorsal STG), and higher-level combinatorial processes (anterior temporal/inferior frontal regions). Additional circuits have been delineated that support auditory-motor integration and auditory/phonological short-term memory. Here are some relevant reviews of this literature: Binder et al., 2000; Hickok and Poeppel, 2007; Indefrey and Levelt, 2004.
Regarding question two, several studies have clearly identified voice-specific responses in the STG (Belin et al. 2000), higher-level speech specific responses in the STS (Scott et al. 2000; Okada et al. 2010), and even what we might as well call the anterior temporal lobe sentence area, given how selective it is to the perception of sentence-level stimulation (Humphries et al., 2006; Humphries et al., 2005; Humphries et al., 2001; Rogalsky and Hickok, 2009; Vandenberghe et al., 2002). But more importantly, some of us have moved beyond the specificity issue with the aim of trying to identify the circuits and computations involved in a given process whether or not it is special to speech.
So F&K are a bit misinformed in regarding the contribution of neuroimaging to the questions they raise.
Even worse, though, is their summary of "where things stand" concerning our understanding of the "neural basis of language" -- a rather sweeping domain, especially if they have in mind only the two questions they raise at the outset. Nonetheless, concerning the Neural Basis of Language they emphasize:
1. that the 19th century idea that Broca's area = speech production and Wernicke's area = speech comprehension doesn't hold up to modern data
2. that left frontal regions activate to a variety of language tasks, and indeed even non-linguistic tasks
3. that regions outside of the traditional peri-Sylvian cortex activate during language processing
4. that meta analyses show lots of overlap between language tasks
This is frankly a pathetic summary of the state of the field and a pretentious starting point for the methodological schooling that F&K provide in the following sections of their paper.
Completely ignored in this summary is (i) a body of work showing that much of the confusion (and overlapping activations) evaporates if one is careful about task selection (Hickok & Poeppel, 2007), (ii) convergence on the involvement of the STS in phonemic level processes in speech perception (Leibenthal et al., 2005; Scott & Johnsrude, 2003; Hickok & Poeppel, 2007), (iii) convergence on the idea of a dual stream architecture in language system (Hickok & Poeppel, 2007; Rauchecker & Scott, 2009), (iv) recent progress in mapping the circuit that supports sensory-motor integration in speech processing (Golfinopoulus et al. 2009; Hickok et al., 2009), (v) progress in understanding the basis of hemispheric asymmetries for acoustic and phonemic processing in auditory cortex (Boemio, et al. 2005); Zatorre, et al. 2002) , (vi) convergence on the idea that anterior temporal regions support some aspect of sentence-level processing (the linguistic equivalent of the FFA), (vii) convergence on the relation between sensory-motor circuits and phonological short-term memory (Buchsbaum et al. 2008; Postle, 2006)... I could go on.
Yes, there is still plenty of murkiness, much of it surrounding the function of Broca's area, and yes, individual subject analyses would be helpful, but it is not a magic bullet (e.g., task selection is more important in my view) -- e.g., I'm willing to bet that the vision folks still have some work to do -- and the existence of murkiness doesn't justify the characterization of an entire field as failing to make progress due to methodological ineptness. This kind of argumentation was prominent in another of Fedorencko's papers that was featured prominently on this blog. It's a bit disturbing to see it showing up again.
F&K's paper has generated a more formal (i.e., published) response by Grodzinsky (2010) who is critical of their take on the field as well but for different reasons. Definitely worth a look.
The field of the neural basis of language has made significant progress in the last several years, despite what F&K assert.
References
Fedorenko, E., & Kanwisher, N. (2009). Neuroimaging of Language: Why Hasn't a Clearer Picture Emerged? Language and Linguistics Compass, 3 (4), 839-865 DOI: 10.1111/j.1749-818X.2009.00143.x
Binder, J.R., Frost, J.A., Hammeke, T.A., Bellgowan, P.S., Springer, J.A., Kaufman, J.N., and Possing, E.T. (2000). Human temporal lobe activation by speech and nonspeech sounds. Cerebral Cortex 10, 512-528.
Belin, P., Zatorre, R.J., Lafaille, P., Ahad, P., and Pike, B. (2000). Voice-selective areas in human auditory cortex. Nature 403, 309-312.
Buchsbaum, B.R., and D'Esposito, M. (2008). The search for the phonological store: from loop to convolution. J Cogn Neurosci 20, 762-778.
Golfinopoulos, E., Tourville, J.A., and Guenther, F.H. (2009). The integration of large-scale neural network modeling and functional brain imaging in speech motor control. Neuroimage.
Grodzinsky, Yosef. 2010. A clearer view of the linguistic brain: reply to Fedorenko and Kanwisher. Language and Linguistic Compass, 4, pp. 605-622.
Hickok, G., and Poeppel, D. (2007). The cortical organization of speech processing. Nat Rev Neurosci 8, 393-402.
Humphries, C., Binder, J.R., Medler, D.A., and Liebenthal, E. (2006). Syntactic and semantic modulation of neural activity during auditory sentence comprehension. J Cogn Neurosci 18, 665-679.
Humphries, C., Love, T., Swinney, D., and Hickok, G. (2005). Response of anterior temporal cortex to syntactic and prosodic manipulations during sentence processing. Human Brain Mapping 26, 128-138.
Humphries, C., Willard, K., Buchsbaum, B., and Hickok, G. (2001). Role of anterior temporal cortex in auditory sentence comprehension: An fMRI study. Neuroreport 12, 1749-1752.
Indefrey, P., and Levelt, W.J. (2004). The spatial and temporal signatures of word production components. Cognition 92, 101-144.
Liebenthal, E., Binder, J.R., Spitzer, S.M., Possing, E.T., and Medler, D.A. (2005). Neural substrates of phonemic perception. Cereb Cortex 15, 1621-1631.
Okada, K., and Hickok, G. (2006). Identification of lexical-phonological networks in the superior temporal sulcus using fMRI. Neuroreport 17, 1293-1296.
Okada, K., Rong, F., Venezia, J., Matchin, W., Hsieh, I.H., Saberi, K., Serences, J.T., and Hickok, G. (in press). Hierarchical Organization of Human Auditory Cortex: Evidence from Acoustic Invariance in the Response to Intelligible Speech. Cereb Cortex.
Postle, B.R. (2006). Working memory as an emergent property of the mind and brain. Neuroscience 139, 23-38.
Rauschecker, J.P., and Scott, S.K. (2009). Maps and streams in the auditory cortex: nonhuman primates illuminate human speech processing. Nat Neurosci 12, 718-724.
Rogalsky, C., and Hickok, G. (2009). Selective Attention to Semantic and Syntactic Features Modulates Sentence Processing Networks in Anterior Temporal Cortex. Cereb Cortex 19, 786-796.
Scott, S.K., and Johnsrude, I.S. (2003). The neuroanatomical and functional organization of speech perception. Trends Neurosci 26, 100-107.
Scott, S.K., Blank, C.C., Rosen, S., and Wise, R.J.S. (2000). Identification of a pathway for intelligible speech in the left temporal lobe. Brain 123, 2400-2406.
Vandenberghe, R., Nobre, A.C., and Price, C.J. (2002). The response of left temporal cortex to sentences. J Cogn Neurosci 14, 550-560.
Friday, August 20, 2010
Conduction aphasia, speech repetition, and the left parietal lobe
Julius Fridriksson has been featured on this blog before and now his team has just published another noteworthy paper in J. Neuroscience. This paper sought to identify the neural correlate of repetition disorder in aphasia. Repetition deficits are characteristic of conduction aphasia although they are not exclusive to conduction aphasia nor is repetition the only deficit in conduction aphasia.
Some historical background is useful, if for no other reason than most people get it wrong in one way or another. Here are two myths/misunderstandings about conduction aphasia.
1. It is a disorder of repetition.
2. It was first discovered/reported by Lichtheim.
As Fridricksson is careful to point out, the repetition disorder is only one symptom of conduction aphasia, the others being impaired word-finding and phonemic paraphasias in production. In fact, the first characterization of the symptoms of conduction aphasia (by Wernicke not Lichtheim) did not make reference to a repetition deficit; rather the hallmarks were impaired word-finding and speech errors, typically with many self-corrective attempts, in the face of otherwise fluent and grammatical speech and good comprehension. Describing the predicted theoretical consequences of a lesion to the connecting pathway between the sensory and motor speech centers, Wernicke (1874) writes,
Repetition was a clinical assessment invented by Lichtheim which essentially was a means to highlight the paraphasic deficit. In my view, conduction aphasia is a disorder of speech production affecting phonological/phonetic levels of processing and which manifests in paraphasic output on a range of tasks including naming, connected speech and repetition.
Regarding the second point, Wernicke was the first to describe a case of conduction aphasia. Most people get this wrong as the Fridriksson et al. paper demonstrates, "Although Wernicke had never seen such a patient, one was later described by Lichtheim" (p. 11057). Not that it matters that much to the science, but it is nice to get the history straight, so here is a quote from Wernicke 1874 (p. 73-74), 11 year before Lichtheim's monograph appeared:
Classically, conduction aphasia is thought to result from a disconnection between sensory and motor speech areas caused by damage to the arcuate fasciculus (the idea of AF involvement came after Wernicke's 1874 monograph, but he bought into it). Recent work has provided a pretty strong case against the AF being the critical structure. Here's a previous entry on this topic based on the work of Nina Dronkers and colleagues.
Ok, enough with the history. Now on to the paper. Friderksson et al. studied a series of 45 acute stroke patients behaviorally, including a test of repetition, and neuroradiologically. For the latter they acquired by structural MRI and perfusion weighted MRI. The use of perfusion weighted imaging in acute stroke is a method championed by Argye Hills at Johns Hopkins, and is, in my view, an excellent tool.
What's interesting about the study is that they didn't select patients on the basis of aphasia type or even the presence of a repetition disorder. Instead they included a range of patients, measured their repetition ability and looked to see what correlated with deficits. This is a useful approach. The only problem in this case is that, as noted in the paper, repetition deficits can result from disruption anywhere along the pathway between perception and production (e.g., peripheral hearing loss will cause a repetition deficit), so in this sense the study is kind of a shotgun approach that will only capture central tendencies. Nonetheless, here is what they found.
Structural damage to the white matter beneath the left supramarginal gyrus, which includes the arcuate fasciculus, was the most strongly correlated region with repetition impairment. HOWEVER, perfusion imaging told a different story, implicating a cortical zone that included the parietal operculum (inferior SMG) (see their Figure 2, bottom row) and a temporal-parietal junction region (which unfortunately they don't picture). This is the same general region implicated in conduction aphasia (Baldo et al. 2008) and where sensory-motor area Spt lives (Hickok et al. 2003, 2009).
Fridriksson et al. are appropriately cautious in concluding that it is the cortical involvement that causes the deficit, instead concluding that speech repetition is "strongly associated with damage to the left arcuate fasciculus, supramarginal gryus, and TPJ" (p. 11060). I wouldn't disagree that the AF, as a connecting pathway plays an important role, but I would argue strongly that the deficit results, computationally speaking, from damage to cortex, area Spt in particular.
In addition, Fridriksson et al. suggest that their findings do not address the other symptoms of conduction aphasia. It is true that they didn't explicitly examine these symptoms, but I believe the symptoms are connected, particularly the repetition and phonemic paraphasias.
References
Baldo JV, Klostermann EC, & Dronkers NF (2008). It's either a cook or a baker: patients with conduction aphasia get the gist but lose the trace. Brain and language, 105 (2), 134-40 PMID: 18243294
Fridriksson J, Kjartansson O, Morgan PS, Hjaltason H, Magnusdottir S, Bonilha L, & Rorden C (2010). Impaired speech repetition and left parietal lobe damage. The Journal of neuroscience : the official journal of the Society for Neuroscience, 30 (33), 11057-61 PMID: 20720112
Hickok, G., Buchsbaum, B., Humphries, C., & Muftuler, T. (2003). Auditory-Motor Interaction Revealed by fMRI: Speech, Music, and Working Memory in Area Spt Journal of Cognitive Neuroscience, 15 (5), 673-682 DOI: 10.1162/jocn.2003.15.5.673
Hickok G, Okada K, & Serences JT (2009). Area Spt in the human planum temporale supports sensory-motor integration for speech processing. Journal of neurophysiology, 101 (5), 2725-32 PMID: 19225172
Wernicke, C. (1874/1969). The symptom complex of aphasia: A psychological study on an anatomical basis. In Boston studies in the philosophy of science, R.S. Cohen, and M.W. Wartofsky, eds. (Dordrecht: D. Reidel Publishing Company), pp. 34-97.
Some historical background is useful, if for no other reason than most people get it wrong in one way or another. Here are two myths/misunderstandings about conduction aphasia.
1. It is a disorder of repetition.
2. It was first discovered/reported by Lichtheim.
As Fridricksson is careful to point out, the repetition disorder is only one symptom of conduction aphasia, the others being impaired word-finding and phonemic paraphasias in production. In fact, the first characterization of the symptoms of conduction aphasia (by Wernicke not Lichtheim) did not make reference to a repetition deficit; rather the hallmarks were impaired word-finding and speech errors, typically with many self-corrective attempts, in the face of otherwise fluent and grammatical speech and good comprehension. Describing the predicted theoretical consequences of a lesion to the connecting pathway between the sensory and motor speech centers, Wernicke (1874) writes,
In this case... the patient understands everything. He can always express himself, but his ability to choose the correct word is disturbed in a manner similar to that in the form just described [sensory, aka Wernicke's aphasia]. p. 54
Repetition was a clinical assessment invented by Lichtheim which essentially was a means to highlight the paraphasic deficit. In my view, conduction aphasia is a disorder of speech production affecting phonological/phonetic levels of processing and which manifests in paraphasic output on a range of tasks including naming, connected speech and repetition.
Regarding the second point, Wernicke was the first to describe a case of conduction aphasia. Most people get this wrong as the Fridriksson et al. paper demonstrates, "Although Wernicke had never seen such a patient, one was later described by Lichtheim" (p. 11057). Not that it matters that much to the science, but it is nice to get the history straight, so here is a quote from Wernicke 1874 (p. 73-74), 11 year before Lichtheim's monograph appeared:
The following is a clear case of conduction aphasia... He understands everything correctly and always answers questions correctly. ... He shows no trace of motor aphasia .... He cannot, however, find words for many objects he wishes to designate [word finding deficit]. He makes an effort to find them, becoming agitated in the process, and if one names them for him he repeats the name without hesitation. ... He can say many things fluently, especially familiar expressions. He then comes to a word on which he stumbles, remains caught on it, exerts himself and becomes irritated. After that every word that he utters, haltingly, is nonsensical [phonemic paraphasias]; he corrects himself over and over again [self-corrective attempts], and the harder he tries the worse the situation becomes...
Classically, conduction aphasia is thought to result from a disconnection between sensory and motor speech areas caused by damage to the arcuate fasciculus (the idea of AF involvement came after Wernicke's 1874 monograph, but he bought into it). Recent work has provided a pretty strong case against the AF being the critical structure. Here's a previous entry on this topic based on the work of Nina Dronkers and colleagues.
Ok, enough with the history. Now on to the paper. Friderksson et al. studied a series of 45 acute stroke patients behaviorally, including a test of repetition, and neuroradiologically. For the latter they acquired by structural MRI and perfusion weighted MRI. The use of perfusion weighted imaging in acute stroke is a method championed by Argye Hills at Johns Hopkins, and is, in my view, an excellent tool.
What's interesting about the study is that they didn't select patients on the basis of aphasia type or even the presence of a repetition disorder. Instead they included a range of patients, measured their repetition ability and looked to see what correlated with deficits. This is a useful approach. The only problem in this case is that, as noted in the paper, repetition deficits can result from disruption anywhere along the pathway between perception and production (e.g., peripheral hearing loss will cause a repetition deficit), so in this sense the study is kind of a shotgun approach that will only capture central tendencies. Nonetheless, here is what they found.
Structural damage to the white matter beneath the left supramarginal gyrus, which includes the arcuate fasciculus, was the most strongly correlated region with repetition impairment. HOWEVER, perfusion imaging told a different story, implicating a cortical zone that included the parietal operculum (inferior SMG) (see their Figure 2, bottom row) and a temporal-parietal junction region (which unfortunately they don't picture). This is the same general region implicated in conduction aphasia (Baldo et al. 2008) and where sensory-motor area Spt lives (Hickok et al. 2003, 2009).
Fridriksson et al. are appropriately cautious in concluding that it is the cortical involvement that causes the deficit, instead concluding that speech repetition is "strongly associated with damage to the left arcuate fasciculus, supramarginal gryus, and TPJ" (p. 11060). I wouldn't disagree that the AF, as a connecting pathway plays an important role, but I would argue strongly that the deficit results, computationally speaking, from damage to cortex, area Spt in particular.
In addition, Fridriksson et al. suggest that their findings do not address the other symptoms of conduction aphasia. It is true that they didn't explicitly examine these symptoms, but I believe the symptoms are connected, particularly the repetition and phonemic paraphasias.
References
Baldo JV, Klostermann EC, & Dronkers NF (2008). It's either a cook or a baker: patients with conduction aphasia get the gist but lose the trace. Brain and language, 105 (2), 134-40 PMID: 18243294
Fridriksson J, Kjartansson O, Morgan PS, Hjaltason H, Magnusdottir S, Bonilha L, & Rorden C (2010). Impaired speech repetition and left parietal lobe damage. The Journal of neuroscience : the official journal of the Society for Neuroscience, 30 (33), 11057-61 PMID: 20720112
Hickok, G., Buchsbaum, B., Humphries, C., & Muftuler, T. (2003). Auditory-Motor Interaction Revealed by fMRI: Speech, Music, and Working Memory in Area Spt Journal of Cognitive Neuroscience, 15 (5), 673-682 DOI: 10.1162/jocn.2003.15.5.673
Hickok G, Okada K, & Serences JT (2009). Area Spt in the human planum temporale supports sensory-motor integration for speech processing. Journal of neurophysiology, 101 (5), 2725-32 PMID: 19225172
Wernicke, C. (1874/1969). The symptom complex of aphasia: A psychological study on an anatomical basis. In Boston studies in the philosophy of science, R.S. Cohen, and M.W. Wartofsky, eds. (Dordrecht: D. Reidel Publishing Company), pp. 34-97.
Wednesday, August 11, 2010
SoCal Hearing Meeting -- UC Irvine
For those of you in the So. Cal. area, this should be a fun meeting...
The Center for Hearing Research at UC Irvine will host an informal one-day conference intended to enhance communication among Southern California scientists interested in hearing. The date is Friday, September 17, 2010. All levels of researchers are welcome, students and staff through faculty, but we particularly would like to encourage presentations from senior students and post-docs. The event will consist of 15-min talks (plus 5 minutes for questions), no more than one from each lab, as well as posters from anyone who would like to offer one. We hope that each speaker will take the first few minutes to briefly introduce the overall scope of research in his or her lab followed by a presentation of a specific research result. If it happens that we receive more volunteers for talks than we can accommodate, we will be back in touch with you to try to trim things down to one or two talks per institution. The posters can be new, but repeats from recent ARO or other meetings also are welcome. We will provide lunch and coffee and snacks throughout the day. We also will organize an optional dinner at a local restaurant, the cost of dinner to be covered by each diner. Aside from the $8.00 parking fee and the cost of the optional dinner, there will be no fee. We do, however, request that you RSVP through the website so that we know how many participants to expect.
Information and registration is at: http://hss.bio.uci.edu/socal_conf.htm
Tentative schedule:
8:30 Welcome, with coffee and bagels
9 to 10:20 4 talks
10:20 to 10:50 break, poster viewing, coffee and snacks
10:50 to 12:10 4 talks
12:10-2 lunch and poster viewing
2-3:20 4 talks
3:20-3:50 break, poster viewing, coffee and snacks
3:50-5:10 4 talks
5:10 Adjourn; optional dinner at a local restaurant
We hope that you can attend.
John Middlebrooks and Virginia Richards
Conference co-organizers
The Center for Hearing Research at UC Irvine will host an informal one-day conference intended to enhance communication among Southern California scientists interested in hearing. The date is Friday, September 17, 2010. All levels of researchers are welcome, students and staff through faculty, but we particularly would like to encourage presentations from senior students and post-docs. The event will consist of 15-min talks (plus 5 minutes for questions), no more than one from each lab, as well as posters from anyone who would like to offer one. We hope that each speaker will take the first few minutes to briefly introduce the overall scope of research in his or her lab followed by a presentation of a specific research result. If it happens that we receive more volunteers for talks than we can accommodate, we will be back in touch with you to try to trim things down to one or two talks per institution. The posters can be new, but repeats from recent ARO or other meetings also are welcome. We will provide lunch and coffee and snacks throughout the day. We also will organize an optional dinner at a local restaurant, the cost of dinner to be covered by each diner. Aside from the $8.00 parking fee and the cost of the optional dinner, there will be no fee. We do, however, request that you RSVP through the website so that we know how many participants to expect.
Information and registration is at: http://hss.bio.uci.edu/socal_conf.htm
Tentative schedule:
8:30 Welcome, with coffee and bagels
9 to 10:20 4 talks
10:20 to 10:50 break, poster viewing, coffee and snacks
10:50 to 12:10 4 talks
12:10-2 lunch and poster viewing
2-3:20 4 talks
3:20-3:50 break, poster viewing, coffee and snacks
3:50-5:10 4 talks
5:10 Adjourn; optional dinner at a local restaurant
We hope that you can attend.
John Middlebrooks and Virginia Richards
Conference co-organizers
Importance of phonemes in speech production
In a previous post I have questioned whether we need to explicitly represent phonemes in speech perception. Massaro and others have raised this issue in the past. Phonemes, the line of thinking goes, are only really important for production. There are linguistic arguments for this that I won't detail here. There is also well-known speech error data which shows that phoneme size units can break off and dislocate themselves. Here I want to highlight some evidence from aphasia. A reviewer of one of my papers pointed me to this study by Lindsey Nickels and David Howard.
A group of aphasics who exhibited speech production errors were asked to repeat words that varied in terms of the number of phonemes, number of syllables, or syllable complexity (defined in terms of consonant clusters). These variables are, of course, highly correlated, but the stimuli were carefully designed so that the contribution of each of these factors could be examined using logistic regression analyses.
The main result was that only number of phonemes in a word predicted correct repetition (see graph below derived from their Table 4) and once this variable was taken into account, the number of syllables or syllable complexity did not explain any additional variance.

Phonemes seem to matter in speech production. I have to say, though, that I'm not fully convinced that the others factors aren't also important.
Nickels, L., & Howard, D. (2004). Dissociating Effects of Number of Phonemes, Number of Syllables, and Syllabic Complexity on Word Production in Aphasia: It's the Number of Phonemes that Counts Cognitive Neuropsychology, 21 (1), 57-78 DOI: 10.1080/02643290342000122
A group of aphasics who exhibited speech production errors were asked to repeat words that varied in terms of the number of phonemes, number of syllables, or syllable complexity (defined in terms of consonant clusters). These variables are, of course, highly correlated, but the stimuli were carefully designed so that the contribution of each of these factors could be examined using logistic regression analyses.
The main result was that only number of phonemes in a word predicted correct repetition (see graph below derived from their Table 4) and once this variable was taken into account, the number of syllables or syllable complexity did not explain any additional variance.

Phonemes seem to matter in speech production. I have to say, though, that I'm not fully convinced that the others factors aren't also important.
Nickels, L., & Howard, D. (2004). Dissociating Effects of Number of Phonemes, Number of Syllables, and Syllabic Complexity on Word Production in Aphasia: It's the Number of Phonemes that Counts Cognitive Neuropsychology, 21 (1), 57-78 DOI: 10.1080/02643290342000122
Monday, August 2, 2010
A robotic model of the human vocal tract?
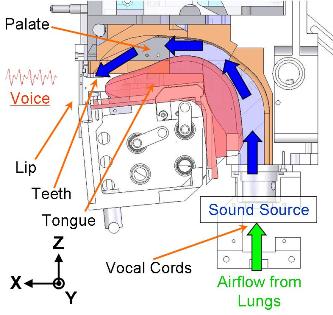
It's not pretty, but this anthropomorphic bot developed at Waseda University in the Takanishi lab can talk -- well, produce some vowels anyway. Have a look here.

Subscribe to:
Posts (Atom)